By Kimberlee Blevins

Introduction
Increasing human population and associated industrial growth have contributed to levels of unsustainability in natural resources (Eilers et al. 2011). These issues are beginning to cause harm to all biodiversity and the ability to balance ecosystem services(Kremen et al. 2002; Greenleaf and Kremen 2006). Insect pollination promotes ecosystem biodiversity and serves as an ecosystem service (Eilers et al. 2011). Farmers are reliant on pollinators for crop production (Free 1993), but insect pollinators face hardships such as: urban sprawl, climate change, insecticides, and other damaging effects (Kearns et al.1998). Human nutrition relies heavily on pollinated crops; thus, pollinators directly impact food security and human health (Eilers et al. 2011).
Insect pollinators are composed of bees, wasps, flies, beetles, butterflies, along with other various insects (Free 1993). These insect pollinators are vital for crops such as fruits, vegetables, nut and seed crops (Free 1993). According to Gallai et al. (2009), “the global economic value of wild and managed pollination services was estimated at $215 billion in 2005, representing 9.5% of the overall global food production.” Previous studies indicate that there is a shortage of pollinators to meet supply and demand for crops (Gallai et al. 2009). This shortage is becoming more alarming as wild insect pollinator populations are declining, while other species are becoming endangered and extinct (Biesmeijer et al. 2006). For many locations, agriculture and increasing urban sprawl have low pollinator outputs that are not meeting demand including species like the honey bee and other wild pollinators (Gallai et al. 2009). The insect-pollinator decline issue demonstrates a need to re-evaluate the pollinator problem and to address artificial pollination tactics (Gallai et al. 2009).
Pollinating species provide enormous ecological and economic benefits to flowering plants, wildlife, and humans (Eilers et al. 2011). Wild bees are large contributors to the pollination of different crops (Kremen et al. 2002; Gallai et al. 2009). As bees are becoming critical pollinators, the focus has been on the introduced bee species, the honey bee (Apis mellifera). Although, the honey bee is the primary focus of the insect pollinator decline, the solitary bee is another important pollinator that provides adequate pollination and receives less attention. (Eilers et al. 2011).
The alfalfa leafcutter bee (ALB) is the most managed solitary bee (Pitt-Singer and Cane 2011). THE ALB was first introduced to the US in the 1930s and was used to boost alfalfa crop yields (Pitt-Singer and Cane 2011). Pitt-Singer and Cane (2011) stated that “the value of the alfalfa leafcutter bee, Megachile rotundata, is surpassed only by the honey bee for pollination of field crops.” Pestilence and unexplained mortality affect US ALB populations at an increasingly higher rate in comparison with Canada populations (Pitt-Singer and Cane 2011). Thus, sustaining ALB populations within the US during the phases of metamorphosis is extremely difficult, and therefore producers choose to import ALBs internationally from Canada (Pitt-Singer and Cane 2011).
As alfalfa leafcutter bees are shipped from Canada to the US, shipping problems arise and can have damaging effects on colony health (Ahn et al 2012; Wyns 2018). These issues are due in part to long haul moves on open freight containers under varying qualities of conditions (Ahn et al 2012; Wyns 2018). Commercial beekeepers are consistently refining their methods to accommodate the problems. (Wyns 2018). Although shipping practices are improving, there are still issues such as tipping over, temperature, ventilation, and other biological stresses (Wyns 2018). A recent concern has been ventilation and oxygen access within the transportation cargo area (Wyns 2018). The confined spaces may limit access to oxygen in some compartments as opposed to other areas that are more ventilated (Wyns 2018).
Insects have a range of adaptations to aid in tolerance to hypoxic conditions within various habitats (Owings 2014). “Data gases sampled underground from tiger beetle larvae burrows reflected levels of O2 as low as 12% whereas decaying stumps were as low as 14%” (Zerm et al. 2004; Benett et al. 2013). Within the adult stage of the M. rotundata, the species has been known to tolerate hypoxia levels well (Benett et al. 2013). The adults maintain metabolic rates at levels of 4% O2, but for other stages during the life cycle, hypoxia tolerance is unknown (Benett et al. 2013).
The ALB is a cavity-nesting insect (Pitts-Singer and Cane 2011; Owings 2014). The eggs go through metamorphosis within brood cells (Pitts-Singer and Cane 2011; Owings 2014) which are created by the female, usually from alfalfa leaves cut in circular disc shapes (Pitts-Singer and Cane 2011; Owings 2014). The brood cells are placed inside beetle tunnels in wood or plant stalks (Pitts-Singer and Cane 2011; Owings 2014). The female then completely seals the brood cells with a paste of leaves (Pitts-Singer and Cane 2011; Owings 2014). Research suggests, the natural brood cell structure may create hypoxic conditions (Pitts-Singer and Cane 2011; Owings 2014). During the beginning stages of the life cycle, pupae and larvae are held compacted within the brood cell for prolonged amounts of time (Owings 2014). At this time of development, ALBs also go through winter and emerge into the adult phase after metamorphosis (Owings 2014). It is difficult to monitor respiratory movements or measure oxygen levels in insects that undergo enclosed conditions, like those found in the brood cell (Anderson and Ultsch 1987; Benett et al. 2013).
However, a study conducted on M. rotundata looked at differences in the developing life cycle stages through variations in respiratory movements, as well as fluctuations in trachea size (Owings 2014). It was suspected that the brood cell may have influenced the oxygen diffusion and therefore attributed to hypoxia (Owings 2014). Critical blood gas levels were measured during the pupae state by measuring CO2 output when subjected to different levels of oxygen (Owings 2014). The study indicated that as pupae aged, hypoxia tolerance decreased (Owings 2014). Owings (2014) concluded that studies focusing on hypoxia within various bee species will aid in contributing to healthy pollinator populations and the bee transport industry.
In the case that M. rotundata are less tolerant to hypoxic conditions as they mature, exposure to lower oxygen levels in late phases of brood cell maturation should result in decreased emergence rates in comparison to those that are exposed to regular oxygen levels. In order to test this hypothesis, two groups of M. rotundata will be run under two conditions, a control group and a treatment group. The control group will be exposed to 21% oxygen, while the treatment group will be exposed to a 10% oxygen level. Post exposure, emergence rates between the two groups will be compared. Assumption are that the bees exposed to a 10% oxygen atmosphere will have a significantly lower emergence rate than those exposed to 21% oxygen atmosphere.
Methods
Alfalfa leafcutter bees were ordered from a laboratory in Logan, UT, while still in the cocoon stage. The cocoons were kept for the length of the project in a single container with a ventilated top in a 29°C incubator. As the cocoons neared adulthood, red-eye pupae and emergence-ready adults were removed from the single container and placed into twenty 24-well plates, one day prior to oxygen treatments. The twenty 24-well plates were then divided evenly into two groups that included group A: the control group and group B: the treatment group. Each well plate in Group A was marked with tape and labeled as 21% oxygen. All the well plates in Group B were marked with tape and labeled as 10% oxygen.
There were ten well-plates for each group. Five well plates from group A were taped down and sealed into a vacuum desiccator. This was repeated for the remainder of group A, as well as group B for a total of four vacuum desiccators. Group A was stored in a 21% oxygen atmosphere that represented a normal atmospheric oxygen concentration. Group B was stored under 10% oxygen atmosphere. To create these conditions, a flow of 6000 ml/min with the correct gas setting of 21% oxygen and 10% oxygen was used. Each vacuum desiccator was run through the prescribed oxygen level for a total of five minutes at the same time every day to maintain the preferred oxygen level. This was done for seven consecutive days.
Between each gassing session, the vacuum desiccators were kept in a =29°C incubator. Each of the four vacuum desiccators were only brought out of the incubator to receive the five-minute gassing session.
At the end of the 7-day period, the brood cell groups were taken from labs in Fargo, ND to Bismarck, ND where the groups were monitored for emergence. Each day every well plate was looked over to watch for a bee to emerge from a brood cell. Upon emergence, the bee’s sex was identified and recorded, along with the date of emergence.
These data were used for the analysis. A Chi-squared test compared expected emergence rates to actual observed rates. The chi-squared test compared treatment females and males to control group females and males. Two one-tailed t-tests assuming equal variances were also performed to compare emergence in the treatment group compared with the control group by gender.
Results
The two groups of the alfalfa leafcutter bees, the control group (21%) and the treatment group (10%), were analyzed using a chi-squared test. The expected emergence rate for the species is a 40:60 ratio in favor of males. The emergence rate from the control group indicated that 45 females emerged in comparison with the males that reached a 67 emergence (Fig. 1). The treatment group reflected a female emergence rate of 44 compared to males that had a count of 62 (Fig.1).
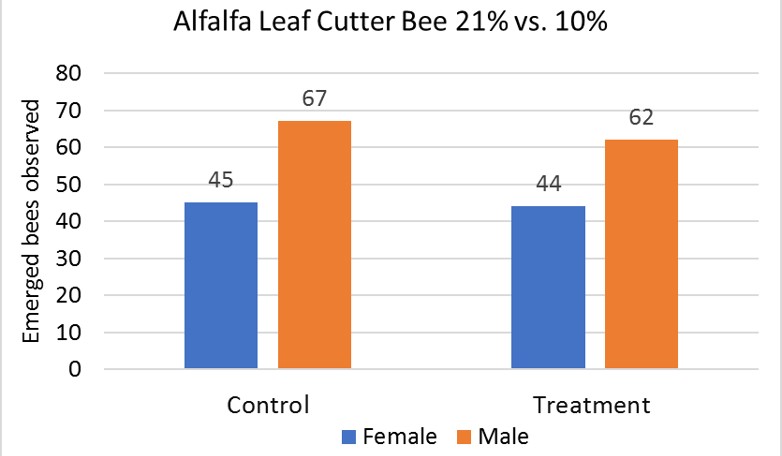
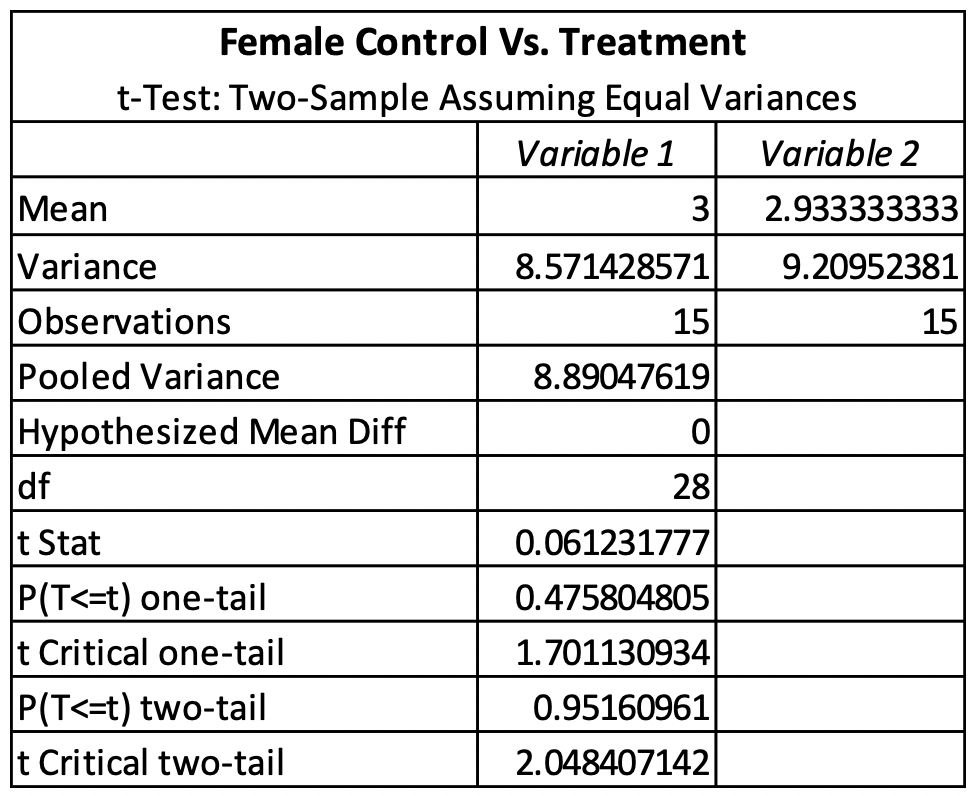
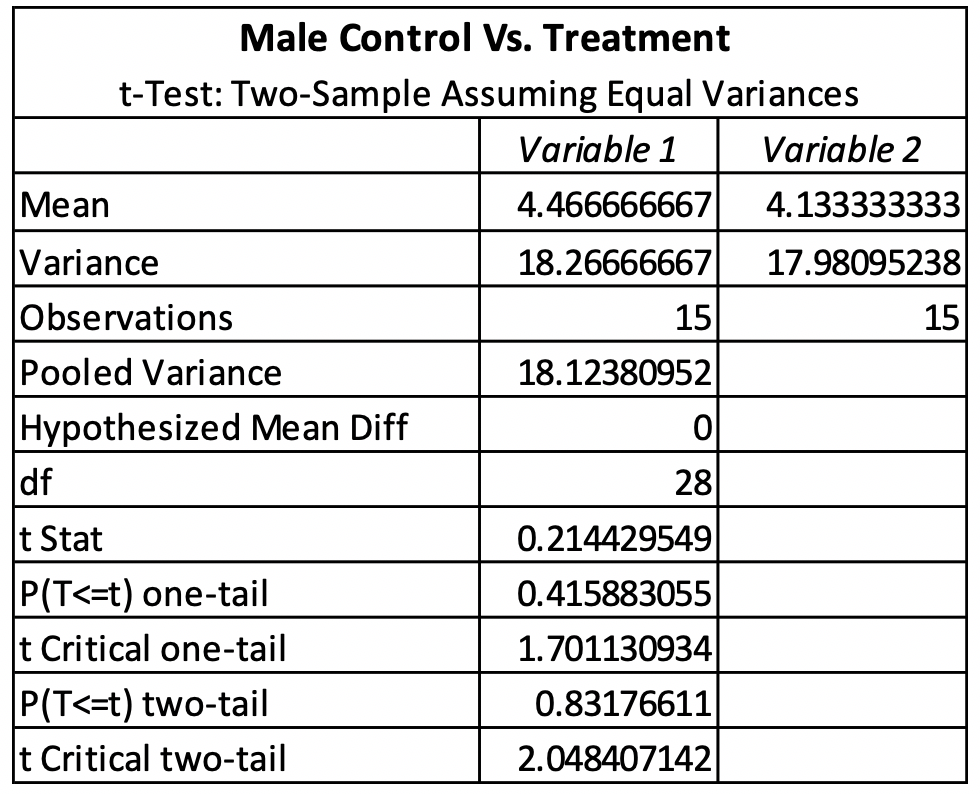
The T-test for female control vs. treatment revealed that there was no significant difference between the control and treatment. The females had a P-value of 0.83176611 (Table 1). The T-test for male control vs. treatment revealed that there was also no significant difference between the control and treatment. The males had a P-value of 0.95160961 (Table 2). The average emergence rate for the female control group was 8.571428571, while the average emergence rate for the female treatment group was 9.20952381 (Table 1). The average emergence rate was also looked at in males. The male control group average emergence rate was 18.26666667, while the average male treatment group for emergence rate was 17.98095238 (Table 2).
Discussion
The alfalfa leafcutter bee is one of the most agriculturally managed pollinators in the US for its production of alfalfa (Pitts-Singer and Cane 2011). Although it is the one of the most managed, there is a problem raising larvae and pupae in the US (Pitts-Singer 2011). Canada has an increasingly higher success rate for raising this species of pollinator (Pitts-Singer 2011). With the high rate of succession in Canada, the species is imported to the US for agricultural purposes (Pitts-Singer 2011). During the transportation of the species, issues arise with the long-haul move (Wyns 2018). Ventilation within the shipping crates is a concern as it may cause low access to oxygen in different spaces of the cargo hold (Wyns 2018).
As the two groups of bees, treatment and control, were analyzed for emergence under two levels of oxygen, it was found that there was no significant difference in the emergence rate of males and females compared between the treatment group, which was 10% oxygen, and the control group that was at 21% oxygen. This would reflect that although there maybe areas of low oxygen access within transport cases, the changes in oxygen level have no significant effect on the rate of emergence. The rate of emergence is of concern if the bees under low oxygen may have a lower emergence rate than those that experience regular atmospheric levels of oxygen (21%). This would ultimately effect pollination success.
During the process of inspecting the bees for emergence from the brood cell, a parasite, Pteromalus venustus, was identified in both the treatment and control groups. The species is referred to as a parasitic wasp and is known to affect the alfalfa leafcutter bee, causing major loss to populations (Whitfield et al. 1985). This parasite brings with it several issues that include an increased number of offspring that it lays within each brood cell of the alfalfa leafcutter bee (Whitfield et al. 1985). The parasite also has a short maturation time giving it the ability to increase very rapidly (Whitfield et al. 1985).
There is no sure method to guarantee 0% rate for parasitism within the brood cells (Goerzen 2011). It is advised for purchases of the ALB to be sampled and broken apart to view for the parasite (Goerzen 2011). This is not always a guarantee as the parasite may still be found within unsampled brood cells (Goerzen 2011). Another form of management is using pyrethrin aerosol, but this insecticide has potential to cause risk to the bee (Goerzen 2011). The contaminated brood cells were removed from the well-plate and set in a separate container. Emergence rates were continued to be monitored for the remaining brood cells in both groups. After emergence monitoring was completed, 50 brood cell samples were randomly chosen from the treatment and control groups. The brood cells were analyzed for parasitic exit holes or cut open to observe for parasitic larvae. Within the treatment group, 36% of the brood cells had parasites present (Fig. 2). The control group had almost double the number of parasites within the brood cells, 58% (Fig 3).
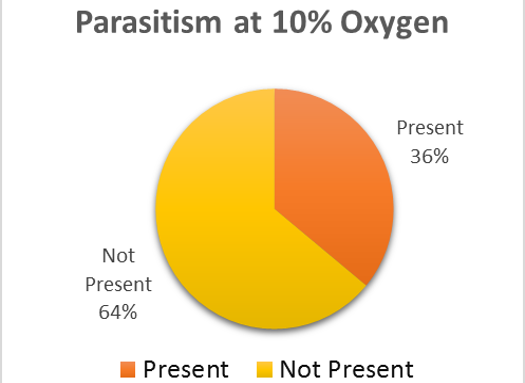
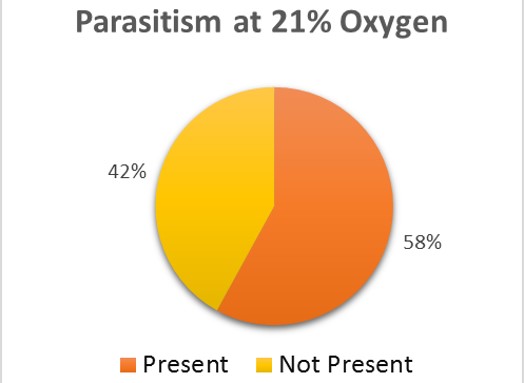
The trend in the data would indicate that low oxygen levels appear to negatively affect the parasite, Pteromalus venustus, but further study would need to substantiate this claim. For further study, the wasp parasite should be analyzed under the same oxygen levels as the alfalfa leafcutter bee with larger sample sizes to further test this theory. If the varying oxygen levels have no significant effect on the emergence of the alfalfa leafcutter bee, this may be a way to have reduced parasitism in the brood cells.
Acknowledgements
Thank you to Courtney Grula and Dr. Julia Bowsher from North Dakota State University for providing guidance and assistance throughout this project. And thank you to United Tribes Technical College and the Environmental Science and Research faculty for providing direction and support in this venture.
References
Ahn K, Xie X, Riddle J, Pettis J, and Huang Z (2012) Effects of Long-Distance Transportation on Honey Bee Physiology. Psyche 9-10.
Anderson J and Ultsch G (1987) Respiratory gas concentrations in the microhabitats of some florida arthropods. Comparative Biochemistry and Physiology Part A: Physiology. 88(3):585-588.
Bennett M, Petersen K, Yocum G, Rinehart J, Kemp W, and Greenlee K (2013)Effects of extended prepupal storage duration on adult flight physiology of the alfalfa leafcutting bee (Megachilidae). Journal of Economic Entomology. 106:1089-1097.
Biesmeijer J, Roberts S, Reemer M, Ohlemüller R, Edwards M, Peeters T, Schaffers A, Potts S (2006) Parallel declines in pollinators and insect-pollinated plants in Britain and the Netherlands. Science 313:351-354.
Eilers E, Kremen C, Greenleaf S, Garber A, Klein A (2011) Contribution of pollinator-mediated crops to nutrients in the human food supply. Public Library of Science 6.
Free J (1993) Insect pollination of crops. Academic Press London.
Gallaia N, Salles J, Settele J, Vaissièrea B (2009) Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecological Economics. 68:810-821.
Greenleaf S, Kremen C (2006) Wild bees enhance honey bees’ pollination of hybrid 15 sunflower. Proceedings of the National Academy of Sciences. 103:13890-13895.
Goerzen D (2011) Parasite control in alfalfa leafcutter bee populations. Saskatchewan Alfalfa Seed Producers Association.
Kearns C, Inouye D, Waser N (1998) Endangered Mutualisms: The Conservation of plant-pollinator interactions. Annual Review of Ecology and Systematics. 29:83-112.
Kremen C, Williams N, and Thorp R (2002) Crop pollination from native bees at risk from agricultural intensification. Proceedings of The National Academy of Sciences. 99:16812-1681.
Owings A, Yocumb G, Rinehart J, Kemp K, and Greenleea K (2014) Changes in respiratory structure and function during post-diapause development in the alfalfa leafcutting bee, Megachile rotundata. Journal of Insect Physiology. 66:20-27.
Pitts-Singer L & Cane J (2011) The Alfalfa Leaf Cutting Bee, Megachile rotundata: The world’s most intensively managed solitary bee. The Annual Review of Entomology. 56:221–37.
Whitfield G and Richards K (1985) Influence of temperature on survival and rate of development of Pteromalus venustus (Hymenoptera: Pteromalidae), a parasite of the alfalfa leafcutter bee (Hymenoptera: Megachilidae). The Canadian Entomology. 117: 811-818.
Wyns D (2018) The long haul. Retrieved from: https://beeinformed.org/2018/02/08/the-long-haul/
Zerm M, Walenciak O, Val A, Adis J (2004) Evidence for anaerobic metabolism in the larval tiger beetle, Phaeoxantha klugii (Col. Cicindelidae) from a Central Amazonian floodplain (Brazil). Physiological Entomology. 29:483-488.
About the Author

Kimberlee Blevins is an enrolled member of the Mandan Hidatsa Arikara Nation. She is a founding member and president of the United Tribes Technical College AISES chapter. Ms. Blevins obtained two associate degrees in pre-engineering and Native American studies and graduated this spring with a bachelor’s in Environmental Science and Research from UTTC. She plans to pursue her master’s in environmental science.
